

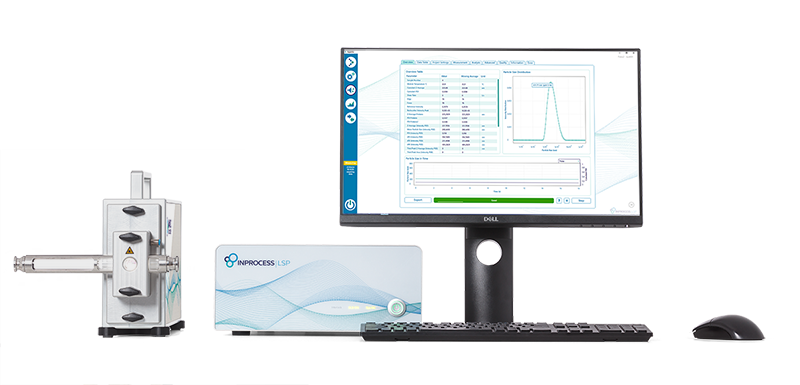
Real-Time Nanoparticle Size Characterization
in Your Process
- Particle size data in seconds
- Continuous monitoring during processing
- Works in high turbidity samples
- Configurable for inline, at-line, online or offline use
FDA-recognized Nanoparticle Analyzer for inline nanoparticle size characterization
What is the NanoFlowSizer?
The NanoFlowSizer is a real-time nanoparticle size analyzer for process environments. Built on Spatially Resolved Dynamic Light Scattering (SR-DLS), it is designed to characterize nanoparticles under process-relevant conditions, including flowing and often highly turbid dispersions.
Unlike conventional offline workflows, the NanoFlowSizer can be integrated inline, online, at-line, or used offline, giving teams faster access to particle size data during formulation, process development, scale-up, and manufacturing.
Typical challenges in nanoparticle size analysis
In many labs and production environments, particle size analysis is still slower, more disruptive, and less representative than it should be.
In practice, this often means:
- Sampling interrupts the process and may break sterility
- Dilution changes the sample before measurement
- Conventional DLS is limited in turbid and dynamic systems
- Offline results often arrive too late for timely process decisions
- Continuous manufacturing needs faster, more robust process feedback

Why teams choose the NanoFlowSizer
The NanoFlowSizer helps you move from delayed offline testing to direct, real-time particle size characterization under process-relevant conditions.
- No dilution required – analyze concentrated and highly turbid samples as they are
- Real-time results – receive updated size data within seconds
- Non-invasive measurement – suitable for closed, sterile, and GMP-aligned processes
- Flexible deployment – use inline, online, at-line, or offline depending on your setup
- Scalable platform – apply the same technology from R&D through manufacturing
- Supports process understanding – detect shifts, aggregation, or out-of-spec trends earlier
Designed for process conditions, not just the lab bench
Traditional DLS methods are valuable for particle characterization, but they are not optimized for complex, in-flow, high-turbidity process conditions. This can constrain their applicability where representative, time-resolved particle size data are required for PAT- and QbD-aligned development and manufacturing.
The NanoFlowSizer is built to bridge that gap. It enables direct measurement in flowing systems and supports faster decisions with less sampling, less manual handling, and better alignment with PAT and QbD strategies.


“Save time, save money, control production processes.”
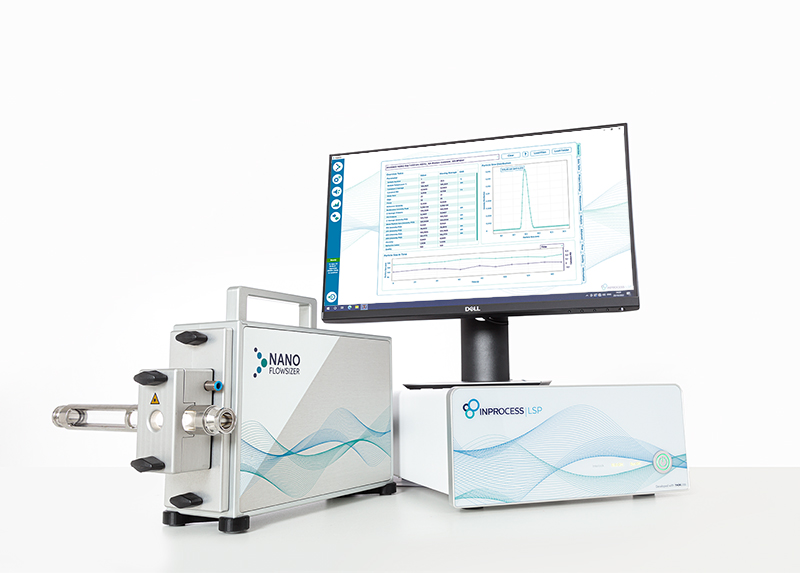
Choose your NanoFlowSizer model
Both NanoFlowSizer models are based on SR-DLS technology and support real-time nanoparticle sizing in flow and static modes. The best choice depends mainly on your sample characteristics and measurement goals.
NanoFlowSizer FIDES
For high-turbidity samples and broad process applicability
FIDES is designed for low to very high turbidity samples and is suitable for both nano- and microparticles. It is a strong fit for applications such as LNPs, inorganic and organic nanoparticles, and milled API suspensions, especially where robust integration into GMP environments is important.
- Designed for high turbidity samples
- Suitable for nano- and microparticles
- Strong fit for GMP-oriented environments
NanoFlowSizer THALIA
For very low turbidity samples and smaller nanoparticles
THALIA is particularly suited for small nanoparticles in very low to high turbidity samples. It is a strong option for protein-based systems and sensitive biological formulations where detection of smaller particles is especially relevant.
- Ideal for very low turbidity samples
- Particularly suited for small nanoparticles
- Strong fit for biologics and protein-related applications
| System feature | FIDES | THALIA |
|---|---|---|
| Measurement technique: | Spatially Resolved DLS (SR-DLS) | Spatially Resolved DLS (SR-DLS) |
| Particle Size Range: | 15 nm – 5000 nm | 3 nm – 5000 nm |
| Turbidity: | Low to very high | Very low to high |
| Measuring mode: | Flow & Static | Flow & Static |
| Configuration: | Inline, online, at-line, offline | Inline, online, at-line, offline |
| Data collection: | Real-time/continuous <5 sec per datapoint | Real-time/continuous <5 sec per datapoint |
| Flow rate: | <250 L/hr | <250 L/hr |
| Nanoparticles: | LNPs, (in)organic particles, milled API | LNPs, (in)organic particles, proteins, mAb, pAb, ADC |
| Software: | XsperGo + GMP compliant, OPC-UA compatible | XsperGo + GMP compliant, OPC-UA compatible |
| Dimensions (probe unit): | 25 * 35 * 10 cm | 25 * 35 * 10 cm |
Where the NanoFlowSizer fits in your workflow
From early formulation work to GMP manufacturing, the NanoFlowSizer supports each process stage with real-time nanoparticle size data.
R&D & Formulation
Support early formulation screening and compare nanoparticle behavior under realistic process conditions.
- Compare formulations faster
- Study particle formation and aggregation
- Generate data that supports later scale-up
- Supports for GMP implementation
Process Development & Scale-Up
Continuous data can reduce repeated offline testing and help build more robust design spaces in line with QbD and PAT principles.
- Monitor the impact of process parameters
- Reduce repeated offline testing
- Support QbD and PAT-driven development
Manufacturing
Monitor particle size and distribution as critical quality attributes during production. React earlier to process deviations.
- Monitor particle size in real time
- Detect drift or aggregation sooner
- Reduce batch-to-batch variability
Typical Application Areas
- Lipid nanoparticles and liposomes
- Protein complexes, biologics, and bioconjugates
- Nano-emulsions
- Milled API suspensions
- Nanoparticle synthesis
- Micelles and polymer nanoparticles

Prefer Offline Analysis?

Meet the NanoLabSizer
For teams working in early R&D, performing batch verification, or preferring benchtop flexibility, the NanoLabSizer offers fast, non-destructive particle size analysis in closed sample containers.
It is built on the same SR-DLS technology as the NanoFlowSizer, but optimized for offline use in vials, syringes, flasks, bags, and other transparent packaging.
Related resources
Ready to implement real-time nanoparticle size characterization in your process?
Replace delayed, offline measurements with faster and more representative particle size data.
Whether you are developing formulations, scaling up a process, or building toward GMP manufacturing, the NanoFlowSizer helps you measure under process-relevant conditions and make decisions with greater confidence.