Lipid-Based Nanoparticles: Manufacturing and inline size Characterization
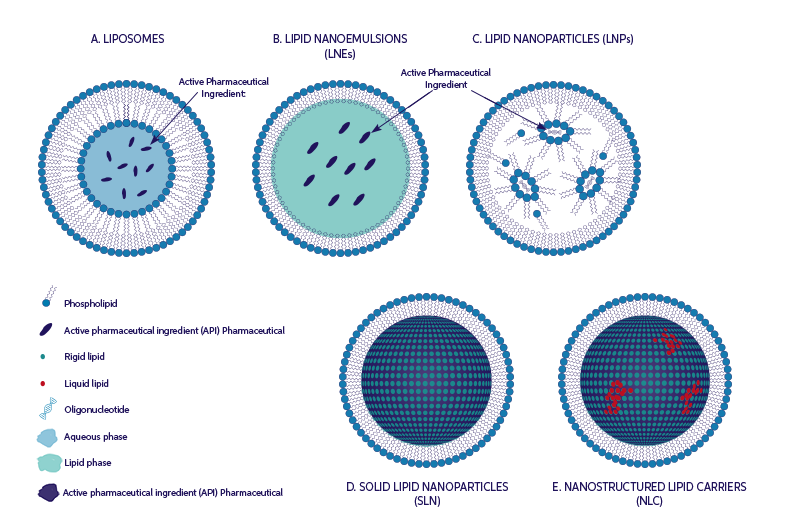
Lipid-based Nano Particles form a very useful class of delivery systems in the pharmaceutical area as well as in cosmetics (and food/nutraceuticals). The recently developed LNP formulations for COVID-19 vaccines highlight this prominent role of Lb-NPs. With the often-complex manufacturing methods of the nano-formulations and the strict size specifications to achieve optimal performance-particularly for nanomedicines-, inline characterization and PAT for nano-suspension manufacturing process has become increasingly important.
The NanoFlowSizer enables for the first time fully non-invasive size characterization in batch and continuous Lb-NPs processes without sample preparation.
Read the full whitepaper:


Up to millions is lost per year due to the absence of appropriate
particle size monitoring solutions…