Liposome manufacturing under continuous flow conditions: towards a fully integrated set-up with in-line control of critical quality attributes
Maryam Sheybanifard, Luis P. B. Guerzoni, Abdolrahman Omidinia-Anarkoli, Laura De Laporte, Johannes Buyel, Rut Besseling, Michiel Damen, Ad Gerich,h Twan Lammers a and Josbert M. Metselaar*
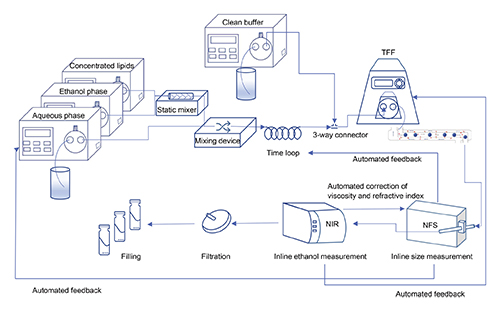
Continuous flow manufacturing (CFM) has shown remarkable advantages in the industrial-scale production of drug-loaded nanomedicines, including mRNA-based COVID-19 vaccines. Thus far, CFM research in nanomedicine has mainly focused on the initial particle formation step, while post-formation production steps are rarely integrated. The opportunity to implement in-line quality control of critical quality attributes merits closer investigation.
Here, we designed and tested a CFM setup for the manufacturing of liposomal nanomedicines that can potentially encompass all manufacturing steps in an end-to-end system. Our main aim was to elucidate the key composition and process parameters that affect the physicochemical characteristics of the liposomes.
Such in-line measurements allow for real-time monitoring and in-process adjustment of key composition and process parameters.
Free download article Sheybanifard et al.:
“Liposome manufacturing under continuous flow conditions: towards a fully integrated set-up with in-line control of critical quality attributes“


“Save time, save money, control production processes.”
Technical Articles
- How do results from different dynamic light scattering instruments compare?
- How can you improve your PEI-based transient transfection process?
- Monitoring Protein Aggregation During Downstream Processing – Insights Using the NanoFlowSizer
- Lipid-Based Nanoparticles: Manufacturing and inline size Characterization