
Real-Time Nanoparticle Size Characterization
No sampling, no dilution. Full process insight.
- Particle size information in seconds
- Continuous size characterization of nanoparticles during processing
- Analysis in high turbidities
- Fully customizable to your process
FDA-recognized Nanoparticle Analyzer for inline nanoparticle size characterization
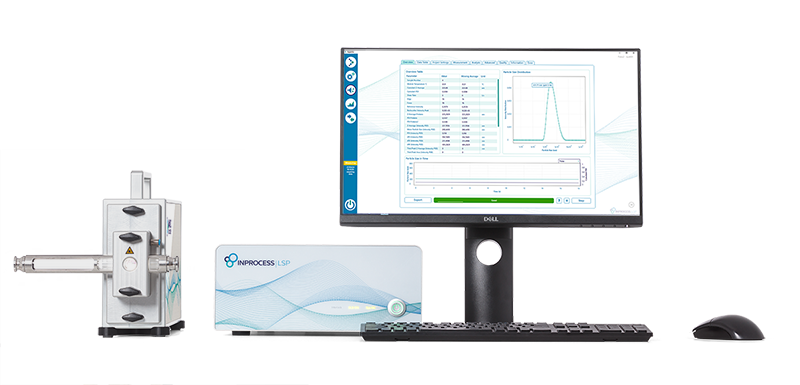
Inline, Continuous Nanoparticle Size Analysis
Discover the NanoFlowSizer
Get fast, reliable particle size information, even in highly turbid samples.
The NanoFlowSizer enables continuous, real-time characterization of nanoparticles during processing; inline, online, at-line, or offline.
Fully configurable to your setup.
In many labs particle size analysis is still:
- Offline – samples are withdrawn and sent to a separate lab
- Time-consuming – dilution, transfer and handling introduce delays and variability
- Disruptive – sampling can affect sterility and process continuity
- Reactive – deviations are detected only after material has already been processed
Do These Challenges Sound Familiar?
- Sampling breaks sterility
- Every measurement requires dilution
- Turbidity limits the use of conventional DLS
- No real-time size control during continuous manufacturing
- You want real-time release and PAT, but lack robust inline tools
- You need a GMP-Ready analyzer

Think DLS, but with superior features
Conventional DLS Holds You Back
Conventional dynamic light scattering (DLS) was never designed for complex, high-turbidity, in-flow systems. It typically requires clear, diluted, static samples measured offline in dedicated cuvettes.
In development and especially in GMP manufacturing, this leads to delays, extra handling steps and sampling bias.
This makes it difficult to truly understand and control nanoparticle processes, especially in continuous or fast-changing environments and in GMP manufacturing, where timely and reliable data are critical.
Where Conventional DLS Stops, the NanoFlowSizer Starts
The NanoFlowSizer is an advanced, GMP-ready nanoparticle size analyzer designed for real-time, non-invasive measurements in flowing, often highly turbid dispersions. It can be operated inline, online, at-line or offline, at flow rates ranging from less than 1 mL/min in R&D setups to over 240 L/h in production environments.
- Inline, non-invasive measurement – no direct product contact, suitable for sterile and GMP processes.
- No dilution – analyze even highly turbid suspensions as-is.
- Real-time results – size and size distribution updates every few seconds.
- GMP-ready scalability – one platform from R&D to GMP manufacturing.


“Save time, save money, control production processes.”
Scalable from Lab to GMP Manufacturing
In contrast to conventional DLS systems, which require static, optically clear samples and dedicated cuvettes, the NanoFlowSizer is specifically designed for flexible integration into real R&D and manufacturing environments, including GMP-regulated facilities. The same SR-DLS platform:
Supports early formulation and process development, extends into pilot and scale-up.
It integrates into GMP manufacturing lines as a PAT tool for routine monitoring and control.
What This Means for Product Quality and Cost
- Prevent batch failures with early size detection
- Cut analysis time from hours to seconds
- Shorten production cycles, no waiting for lab results
- Reduce sampling and lab overhead through fewer manual measurements
- Strengthen regulatory confidence with robust inline nanoparticle analytics that can be implemented in GMP environments and aligned with PAT/QbD strategies.
Typical Application Areas
- Lipid nanoparticles (LNPs), liposomes
- Protein complexes, bioconjugates, biologics
- Nanoemulsions via high-pressure homogenization
- Nanocrystals, milled API suspensions
- Micelles, polymer nanoparticles, self-assembled systems
- Inks, coatings, dispersions

Typical Application Areas
- Lipid nanoparticles (LNPs), liposomes
- Protein complexes, bioconjugates, biologics
- Nanoemulsions via high-pressure homogenization
- Milled API suspensions
- Nanoparticle Synthesis
- Micelles, polymer nanoparticles

NanoFlowSizer Models
| System feature | FIDES | THALIA |
|---|---|---|
| Measurement technique: | Spatially Resolved DLS (SR-DLS) | Spatially Resolved DLS (SR-DLS) |
| Particle Size Range: | 15 nm – 5000 nm | 3 nm – 5000 nm |
| Turbidity*: | Low to very high | Very low to high |
| Measuring mode: | Flow & Static | Flow & Static |
| Configuration: | Inline, online, at-line, offline | Inline, online, at-line, offline |
| Data collection: | Real-time/continuous <5 sec per datapoint | Real-time/continuous <5 sec per datapoint |
| Flow rate: | <250 L/hr | <250 L/hr |
| Nanoparticles: | LNPs, (in)organic particles, milled API | LNPs, (in)organic particles, proteins, mAb, pAb, ADC |
| Software: | XsperGo + GMP compliant, OPC-UA compatible | XsperGo + GMP compliant, OPC-UA compatible |
| Dimensions (probe unit): | 25 * 35 * 10 cm | 25 * 35 * 10 cm |
NanoFlowSizer FIDES
- Designed for high turbidity samples
- Real-time in-flow measurements
- Compatible with nano- and microparticles
- Suitable for integration into GMP environments
NanoFlowSizer THALIA
- Ideal for very low turbidity samples
- Optimized for static measurements
- Particularly suited for small nanoparticles
- Can be used in GMP-supporting labs
Prefer Offline Analysis?

Meet the NanoLabSizer
Whether you’re working in early R&D, need batch verification, or prefer benchtop flexibility, the NanoLabSizer gives you quick, reliable size data in seconds.
The NanoLabSizer is a compact, offline particle size analyzer built on the same SR-DLS technology as the NanoFlowSizer.
It enables fast, dilution-free measurements in transparent containers, no need for flow connections.
Where NanoFlowSizer fits in your workflow
R&D & Formulation
- Rapid screening formulations and process parameters
- Real-time monitoring of nucleation, growth, and aggregation
- Efficient data management
- Ready for future GMP implementation
Process Development & Scale-Up
- Optimize high-pressure homogenization, milling, emulsification and LNP processes with live-size feedback
- Shorten development cycles by replacing repeated offline testing with continuous inline data
- Build robust design spaces in line with QbD and PAT principles
Manufacturing & Commercial Production
- Monitor critical quality attributes (CQAs) like particle size and distribution during production
- Detect drifts, aggregation or out-of-spec trends early, to avoid batch failure
- Reduce batch-to-batch variability and rejection risk, while lowering analytical burden
Ready to implement real-time nanoparticle size process control?
If you’re still:
- Diluting every sample
- Breaking sterility to measure
- Relying on lab results that arrive after critical decisions…
Case Studies & Articles
- How do results from different dynamic light scattering instruments compare?
- How can you improve your PEI-based transient transfection process?
- Monitoring Protein Aggregation During Downstream Processing – Insights Using the NanoFlowSizer
- Lipid-Based Nanoparticles: Manufacturing and inline size Characterization